
2020-4-4 Matter definition: A matter is a task, situation, or event which you have to deal with or think about. Meaning, pronunciation, translations and examples. Log In Dictionary. Dictionary Grammar Blog School Scrabble Thesaurus Translator Quiz More Resources More from Collins. Matter meaning: 1. A situation or subject that is being dealt with or considered: 2. The situation being dealt.
Substance that has mass and volume MatterMatter is usually classified into, with sometimes added as a fourth state. From top to bottom: , , , and a.In and general chemistry, matter is any substance that has and takes up space by having.
This is just a preview! This means war charles jenkins chords. Get the embed code Charles Jenkins & Fellowship Chicago - War (Live) Album Lyrics1.War (Instrumental)2.War (Live)3.War (Radio Version / Live)4.War (Radio Version/Live)5.War - LiveCharles Jenkins & Fellowship Chicago Lyrics provided by SongLyrics.comNote: When you embed the widget in your site, it will match your site's styles (CSS).
All everyday objects that can be touched are ultimately composed of, which are made up of interacting, and in everyday as well as scientific usage, 'matter' generally includes and anything made up of them, and any particles (or ) that act as if they have both and volume. However it does not include such as, or other energy phenomena or waves such as. Matter exists in various (also known as ).
These include classical everyday phases such as, and – for example exists as ice, liquid water, and gaseous steam – but other states are possible, including, and.Usually atoms can be imagined as a of and, and a surrounding 'cloud' of orbiting which 'take up space'. However this is only somewhat correct, because subatomic particles and their properties are governed by their, which means they do not act as everyday objects appear to act – they can act like and they do not have well-defined sizes or positions. In the of, matter is not a fundamental concept because the of atoms are entities which do not have an inherent 'size' or ' in any everyday sense of the word. Due to the and other, some ' known as (, ), and many composites and atoms, are effectively forced to keep a distance from other particles under everyday conditions; this creates the property of matter which appears to us as matter taking up space.For much of the history of the people have contemplated the exact nature of matter. The idea that matter was built of discrete building blocks, the so-called, independently appeared in and among, and in 1st-millennium BC. Ancient philosophers who proposed the particulate theory of matter include (c.
6th–century BC or after), (490 BC) and (470–380 BC). Contents.Comparison with massMatter should not be confused with mass, as the two are not the same in modern physics. Matter is a general term describing any ' physical substance'. By contrast, is not a substance but rather a quantitative property of matter and other substances or systems; various types of mass are defined within – including but not limited to,.While there are different views on what should be considered matter, the mass of a substance has exact scientific definitions. Another difference is that matter has an 'opposite' called antimatter, but mass has no opposite—there is no such thing as 'anti-mass' or, so far as is known, although scientists do discuss the concept. Antimatter has the same (i.e. Positive) mass property as its normal matter counterpart.Different fields of science use the term matter in different, and sometimes incompatible, ways.
Some of these ways are based on loose historical meanings, from a time when there was no reason to distinguish mass from simply a quantity of matter. As such, there is no single universally agreed scientific meaning of the word 'matter'. Scientifically, the term 'mass' is well-defined, but 'matter' can be defined in several ways. Sometimes in the field of physics 'matter' is simply equated with particles that exhibit rest mass (i.e., that cannot travel at the speed of light), such as quarks and leptons. However, in both and, matter exhibits both -like and -like properties, the so-called.
Definition Based on atomsA definition of 'matter' based on its physical and chemical structure is: matter is made up of. Such atomic matter is also sometimes termed ordinary matter. As an example, (DNA) are matter under this definition because they are made of atoms. This definition can be extended to include charged atoms and molecules, so as to include (gases of ions) and (ionic solutions), which are not obviously included in the atoms definition. Alternatively, one can adopt the.Based on protons, neutrons and electronsA definition of 'matter' more fine-scale than the atoms and molecules definition is: matter is made up of what and are made of, meaning anything made of positively charged, neutral, and negatively charged. This definition goes beyond atoms and molecules, however, to include substances made from these building blocks that are not simply atoms or molecules, for example electron beams in an old television, or matter—typically, carbon and oxygen nuclei in a sea of degenerate electrons. At a microscopic level, the constituent 'particles' of matter such as protons, neutrons, and electrons obey the laws of quantum mechanics and exhibit wave–particle duality.
At an even deeper level, protons and neutrons are made up of and the force fields that bind them together, leading to the next definition.Based on quarks and leptons. Under the 'quarks and leptons' definition, the elementary and composite particles made of the (in purple) and (in green) would be matter—while the gauge bosons (in red) would not be matter. However, interaction energy inherent to composite particles (for example, gluons involved in neutrons and protons) contribute to the mass of ordinary matter.As seen in the above discussion, many early definitions of what can be called 'ordinary matter' were based upon its structure or 'building blocks'. On the scale of elementary particles, a definition that follows this tradition can be stated as:'ordinary matter is everything that is composed of and ', or 'ordinary matter is everything that is composed of any elementary fermions except antiquarks and antileptons'. The connection between these formulations follows.Leptons (the most famous being the ), and quarks (of which, such as and, are made) combine to form, which in turn form. Because atoms and molecules are said to be matter, it is natural to phrase the definition as: 'ordinary matter is anything that is made of the same things that atoms and molecules are made of'.
(However, notice that one also can make from these building blocks matter that is not atoms or molecules.) Then, because electrons are leptons, and protons, and neutrons are made of quarks, this definition in turn leads to the definition of matter as being 'quarks and leptons', which are two of the four types of elementary fermions (the other two being antiquarks and antileptons, which can be considered antimatter as described later). Carithers and Grannis state: 'Ordinary matter is composed entirely of particles, namely the up and down quarks, plus the electron and its neutrino.' (Higher generations particles quickly decay into first-generation particles, and thus are not commonly encountered. )This definition of ordinary matter is more subtle than it first appears. All the particles that make up ordinary matter (leptons and quarks) are elementary fermions, while all the are elementary bosons. The that mediate the are not made of quarks or leptons, and so are not ordinary matter, even if they have mass.
In other words, is not something that is exclusive to ordinary matter.The quark–lepton definition of ordinary matter, however, identifies not only the elementary building blocks of matter, but also includes composites made from the constituents (atoms and molecules, for example). Such composites contain an interaction energy that holds the constituents together, and may constitute the bulk of the mass of the composite. As an example, to a great extent, the mass of an atom is simply the sum of the masses of its constituent protons, neutrons and electrons.
However, digging deeper, the protons and neutrons are made up of quarks bound together by gluon fields (see ) and these gluons fields contribute significantly to the mass of hadrons. In other words, most of what composes the 'mass' of ordinary matter is due to the of quarks within protons and neutrons.
For example, the sum of the mass of the three quarks in a is approximately 12.5, which is low compared to the mass of a nucleon (approximately 938 ). The bottom line is that most of the mass of everyday objects comes from the interaction energy of its elementary components.The Standard Model groups matter particles into three generations, where each generation consists of two quarks and two leptons. The first generation is the and quarks, the and the; the second includes the and quarks, the and the; the third generation consists of the and quarks and the. The most natural explanation for this would be that quarks and leptons of higher generations are of the first generations.
If this turns out to be the case, it would imply that quarks and leptons are, rather than.This quark–lepton definition of matter also leads to what can be described as 'conservation of (net) matter' laws—discussed later below. Alternatively, one could return to the mass–volume–space concept of matter, leading to the next definition, in which antimatter becomes included as a subclass of matter.Based on elementary fermions (mass, volume, and space)A common or traditional definition of matter is 'anything that has and (occupies )'. For example, a car would be said to be made of matter, as it has mass and volume (occupies space).The observation that matter occupies space goes back to antiquity. However, an explanation for why matter occupies space is recent, and is argued to be a result of the phenomenon described in the, which applies to. Two particular examples where the exclusion principle clearly relates matter to the occupation of space are white dwarf stars and neutron stars, discussed further below.Thus, matter can be defined as everything composed of elementary fermions. Although we don't encounter them in everyday life, antiquarks (such as the ) and antileptons (such as the ) are the of the quark and the lepton, are elementary fermions as well, and have essentially the same properties as quarks and leptons, including the applicability of the Pauli exclusion principle which can be said to prevent two particles from being in the same place at the same time (in the same state), i.e. Makes each particle 'take up space'.

This particular definition leads to matter being defined to include anything made of these particles as well as the ordinary quark and lepton, and thus also anything made of, which are unstable particles made up of a quark and an antiquark.In general relativity and cosmologyIn the context of, mass is not an additive quantity, in the sense that one can not add the rest masses of particles in a system to get the total rest mass of the system. Thus, in relativity usually a more general view is that it is not the sum of, but the that quantifies the amount of matter. This tensor gives the rest mass for the entire system.
'Matter' therefore is sometimes considered as anything that contributes to the energy–momentum of a system, that is, anything that is not purely gravity. This view is commonly held in fields that deal with such as. In this view, light and other massless particles and fields are all part of 'matter'.StructureIn particle physics, fermions are particles that obey. Fermions can be elementary, like the electron—or composite, like the proton and neutron. In the, there are two types of elementary fermions: quarks and leptons, which are discussed next.Quarks. Main article:Quarks are particles of, implying that they are.
They carry an of − 1⁄ 3 (down-type quarks) or + 2⁄ 3 e (up-type quarks). For comparison, an electron has a charge of −1 e. They also carry, which is the equivalent of the electric charge for the.
Quarks also undergo, meaning that they are subject to the. Quarks are massive particles, and therefore are also subject to.Quark properties namesymbolspinelectric chargemass(/ 2)mass comparable toantiparticleantiparticlesymbolup-type quarksu 1⁄ 2+ 2⁄ 31.5 to 3.3 5 electronsantiupuc 1⁄ 2+ 2⁄ 31160 to 13401 protonanticharmct 1⁄ 2+ 2⁄ 3169,100 to 173,300180 protons or1 atomantitoptdown-type quarksd 1⁄ 2− 1⁄ 33.5 to 6.010 electronsantidownds 1⁄ 2− 1⁄ 370 to 130 200 electronsantistrangesb 1⁄ 2− 1⁄ 34130 to 4370 5 protonsantibottomb. Main article:Baryons are strongly interacting fermions, and so are subject to Fermi–Dirac statistics.
Amongst the baryons are the protons and neutrons, which occur in atomic nuclei, but many other unstable baryons exist as well. The term usually refers to triquarks—particles made of three quarks. Also, 'exotic' baryons made of four quarks and one antiquark are known as, but their existence is not generally accepted.Baryonic matter is the part of the universe that is made of baryons (including all atoms). This part of the universe does not include, or various forms of degenerate matter, such as compose stars. Microwave light seen by (WMAP), suggests that only about 4.6% of that part of the universe within range of the best (that is, matter that may be visible because light could reach us from it), is made of baryonic matter. About 26.8% is dark matter, and about 68.3% is dark energy.As a matter of fact, the great majority of ordinary matter in the universe is unseen, since visible stars and gas inside galaxies and clusters account for less than 10 per cent of the ordinary matter contribution to the mass–energy density of the universe.
Main article:In physics, degenerate matter refers to the ground state of a gas of fermions at a temperature near absolute zero. The requires that only two fermions can occupy a quantum state, one spin-up and the other spin-down.
Hence, at zero temperature, the fermions fill up sufficient levels to accommodate all the available fermions—and in the case of many fermions, the maximum kinetic energy (called the ) and the pressure of the gas becomes very large, and depends on the number of fermions rather than the temperature, unlike normal states of matter.Degenerate matter is thought to occur during the evolution of heavy stars. The demonstration by that have a maximum allowed mass because of the exclusion principle caused a revolution in the theory of star evolution.Degenerate matter includes the part of the universe that is made up of neutron stars and white dwarfs.Strange matter. Main article:Strange matter is a particular form of, usually thought of as a liquid of,.
It is contrasted with, which is a liquid of and (which themselves are built out of up and down quarks), and with non-strange quark matter, which is a quark liquid that contains only up and down quarks. At high enough density, strange matter is expected to be. Strange matter is hypothesized to occur in the core of, or, more speculatively, as isolated droplets that may vary in size from to kilometers.Two meanings of the term 'strange matter'In and, the term is used in two ways, one broader and the other more specific. The broader meaning is just quark matter that contains three flavors of quarks: up, down, and strange.
In this definition, there is a critical pressure and an associated critical density, and when nuclear matter (made of and ) is compressed beyond this density, the protons and neutrons dissociate into quarks, yielding quark matter (probably strange matter). The narrower meaning is quark matter that is more stable than nuclear matter. The idea that this could happen is the 'strange matter hypothesis' of Bodmer and Witten.
In this definition, the critical pressure is zero: the true ground state of matter is always quark matter. The nuclei that we see in the matter around us, which are droplets of nuclear matter, are actually, and given enough time (or the right external stimulus) would decay into droplets of strange matter, i.e.Leptons. Main article:Leptons are particles of, meaning that they are. They carry an of −1 (charged leptons) or 0 e (neutrinos). Unlike quarks, leptons do not carry, meaning that they do not experience the. Leptons also undergo radioactive decay, meaning that they are subject to the.
Leptons are massive particles, therefore are subject to gravity.Lepton propertiesnamesymbolspinelectric chargemass(/ 2)mass comparable toantiparticleantiparticlesymbolcharged leptonse − 1⁄ 2−10.51101 electrone +μ − 1⁄ 2−1105.7 200 electronsantimuonμ +τ − 1⁄ 2−11,777 2 protonsantitauτ +neutrinosνe 1⁄ 20. Phase diagram for a typical substance at a fixed volume. Vertical axis is Pressure, horizontal axis is Temperature.
The green line marks the (above the green line is solid, below it is liquid) and the blue line the (above it is liquid and below it is gas). So, for example, at higher T, a higher P is necessary to maintain the substance in liquid phase. At the the three phases; liquid, gas and solid; can coexist. Above the there is no detectable difference between the phases.
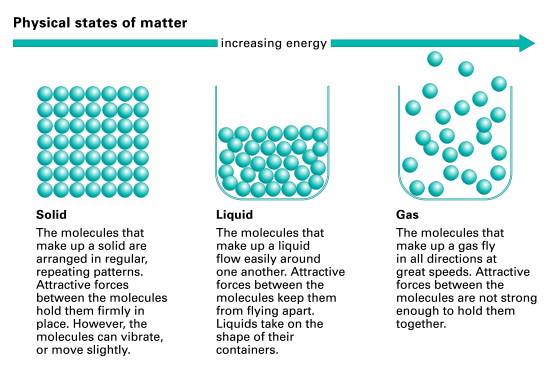
The dotted line shows the: ice melts at constant temperature with increasing pressure.In, matter can exist in several different forms, or states of aggregation, known as, depending on ambient,. A phase is a form of matter that has a relatively uniform chemical composition and physical properties (such as, and so forth). These phases include the three familiar ones (, and ), as well as more exotic states of matter (such as,.). A may be a liquid, gas or plasma. There are also and phases of. As conditions change, matter may change from one phase into another. These phenomena are called, and are studied in the field of.
In nanomaterials, the vastly increased ratio of surface area to volume results in matter that can exhibit properties entirely different from those of bulk material, and not well described by any bulk phase (see for more details).Phases are sometimes called states of matter, but this term can lead to confusion with. For example, two gases maintained at different pressures are in different thermodynamic states (different pressures), but in the same phase (both are gases).Antimatter. Why is there far more matter than antimatter in the observable universe?Antimatter is matter that is composed of the of those that constitute ordinary matter. If a particle and its antiparticle come into contact with each other, the two; that is, they may both be converted into other particles with equal in accordance with 's equation. These new particles may be high-energy or other particle–antiparticle pairs.
The resulting particles are endowed with an amount of kinetic energy equal to the difference between the of the products of the annihilation and the rest mass of the original particle–antiparticle pair, which is often quite large. Depending on which definition of 'matter' is adopted, antimatter can be said to be a particular subclass of matter, or the opposite of matter.Antimatter is not found naturally on Earth, except very briefly and in vanishingly small quantities (as the result of, or ). This is because antimatter that came to exist on Earth outside the confines of a suitable physics laboratory would almost instantly meet the ordinary matter that Earth is made of, and be annihilated. Antiparticles and some stable antimatter (such as ) can be made in tiny amounts, but not in enough quantity to do more than test a few of its theoretical properties.There is considerable speculation both in and as to why the observable universe is apparently almost entirely matter (in the sense of quarks and leptons but not antiquarks or antileptons), and whether other places are almost entirely antimatter (antiquarks and antileptons) instead.
In the early universe, it is thought that matter and antimatter were equally represented, and the disappearance of antimatter requires an asymmetry in physical laws called, which can be obtained from the Standard Model, but at this time the apparent of matter and antimatter in the visible universe is one of the great. Possible processes by which it came about are explored in more detail under.Formally, antimatter particles can be defined by their negative or, while 'normal' (non-antimatter) matter particles have positive baryon or lepton number. These two classes of particles are the antiparticle partners of one another.In October 2017, scientists reported further evidence that matter and, equally produced at the, are identical, should completely annihilate each other and, as a result, the should not exist.
This implies that there must be something, as yet unknown to scientists, that either stopped the complete mutual destruction of matter and antimatter in the early forming universe, or that gave rise to an imbalance between the two forms.Conservation of matterTwo quantities that can define an amount of matter in the quark–lepton sense (and antimatter in an antiquark–antilepton sense), and, are in the Standard Model. A such as the proton or neutron has a baryon number of one, and a quark, because there are three in a baryon, is given a baryon number of 1/3. So the net amount of matter, as measured by the number of quarks (minus the number of antiquarks, which each have a baryon number of −1/3), which is proportional to baryon number, and number of leptons (minus antileptons), which is called the lepton number, is practically impossible to change in any process. Even in a nuclear bomb, none of the baryons (protons and neutrons of which the atomic nuclei are composed) are destroyed—there are as many baryons after as before the reaction, so none of these matter particles are actually destroyed and none are even converted to non-matter particles (like photons of light or radiation). Instead, (and perhaps is released, as these baryons become bound into mid-size nuclei having less energy (and, less compared to the original small (hydrogen) and large (plutonium etc.) nuclei. Even in, there is no net matter being destroyed, because there was zero net matter (zero total lepton number and baryon number) to begin with before the annihilation—one lepton minus one antilepton equals zero net lepton number—and this net amount matter does not change as it simply remains zero after the annihilation.In short, matter, as defined in physics, refers to baryons and leptons.
The amount of matter is defined in terms of baryon and lepton number. Baryons and leptons can be created, but their creation is accompanied by antibaryons or antileptons; and they can be destroyed, by annihilating them with antibaryons or antileptons. Since antibaryons/antileptons have negative baryon/lepton numbers, the overall baryon/lepton numbers aren't changed, so matter is conserved. However, baryons/leptons and antibaryons/antileptons all have positive mass, so the total amount of mass is not conserved.Further, outside of natural or artificial nuclear reactions, there is almost no antimatter generally available in the universe (see and ), so particle annihilation is rare in normal circumstances.Other types. See also: andIn and, dark matter is matter of unknown composition that does not emit or reflect enough electromagnetic radiation to be observed directly, but whose presence can be inferred from gravitational effects on visible matter. Observational evidence of the early universe and the theory require that this matter have energy and mass, but is not composed ordinary baryons (protons and neutrons). The commonly accepted view is that most of the dark matter is.
As such, it is composed of particles as yet unobserved in the laboratory. Perhaps they are, which are not particles, but relics formed at very high energies in the early phase of the universe and still floating about.
See also:In, dark energy is the name given to source of the repelling influence that is accelerating the rate of. Its precise nature is currently a mystery, although its effects can reasonably be modeled by assigning matter-like properties such as energy density and pressure to the itself.Fully 70% of the matter density in the universe appears to be in the form of dark energy. Twenty-six percent is dark matter.
Only 4% is ordinary matter. So less than 1 part in 20 is made out of matter we have observed experimentally or described in the of particle physics.
Of the other 96%, apart from the properties just mentioned, we know absolutely nothing. Main article:Exotic matter is a concept of, which may include dark matter and dark energy but goes further to include any hypothetical material that violates one or more of the properties of known forms of matter. Some such materials might possess hypothetical properties like.Historical development Antiquity (c. 322 BC)In, the Buddhists, the Hindus and the Jains each developed a particulate theory of matter, positing that all matter is made of atoms ( paramanu, pudgala) that are in itself 'eternal, indestructible and innumerable' and which associate and dissociate according to certain fundamental natural laws to form more complex matter or change over time. They coupled their ideas of soul, or lack thereof, into their theory of matter. The strongest developers and defenders of this theory were the Nyaya- school, with the ideas of the philosopher (c. 6th–century BC) being the most followed.
The Buddhists also developed these ideas in late 1st-millennium BCE, ideas that were similar to the Vaishashika Hindu school, but one that did not include any soul or conscience. The Jains included soul ( jiva), adding qualities such as taste, smell, touch and color to each atom.
They extended the ideas found in early literature of the Hindus and Buddhists by adding that atoms are either humid or dry, and this quality cements matter. They also proposed the possibility that atoms combine because of the attraction of opposites, and the soul attaches to these atoms, transforms with residue and transmigrates with each rebirth.In, speculated the underlying nature of the visible world.
546 BC) regarded water as the fundamental material of the world. 546 BC) posited that the basic material was wholly characterless or limitless: the Infinite ( ). (flourished 585 BC, d. 528 BC) posited that the basic stuff was pneuma or air.
475 BC) seems to say the basic element is fire, though perhaps he means that all is change. 490–430 BC) spoke of four of which everything was made: earth, water, air, and fire. Meanwhile, argued that change does not exist, and argued that everything is composed of minuscule, inert bodies of all shapes called atoms, a philosophy called. All of these notions had deep philosophical problems.(384–322 BC) was the first to put the conception on a sound philosophical basis, which he did in his natural philosophy, especially in book I. He adopted as reasonable suppositions the four, but added a fifth,. Nevertheless, these elements are not basic in Aristotle's mind. Rather they, like everything else in the visible world, are composed of the basic principles matter and form.For my definition of matter is just this—the primary substratum of each thing, from which it comes to be without qualification, and which persists in the result.
— Aristotle, Physics I:9:192a32The word Aristotle uses for matter, can be literally translated as wood or timber, that is, 'raw material' for building. Indeed, Aristotle's conception of matter is intrinsically linked to something being made or composed. In other words, in contrast to the early modern conception of matter as simply occupying space, matter for Aristotle is definitionally linked to process or change: matter is what underlies a change of substance. For example, a horse eats grass: the horse changes the grass into itself; the grass as such does not persist in the horse, but some aspect of it—its matter—does. The matter is not specifically described (e.g., as ), but consists of whatever persists in the change of substance from grass to horse. Matter in this understanding does not exist independently (i.e., as a ), but exists interdependently (i.e., as a 'principle') with form and only insofar as it underlies change. It can be helpful to conceive of the relationship of matter and form as very similar to that between parts and whole.
For Aristotle, matter as such can only receive actuality from form; it has no activity or actuality in itself, similar to the way that parts as such only have their existence in a whole (otherwise they would be independent wholes).Seventeenth and eighteenth centuries(1596–1650) originated the modern conception of matter. He was primarily a geometer. Instead of, like Aristotle, deducing the existence of matter from the physical reality of change, Descartes arbitrarily postulated matter to be an abstract, mathematical substance that occupies space:So, extension in length, breadth, and depth, constitutes the nature of bodily substance; and thought constitutes the nature of thinking substance. And everything else attributable to body presupposes extension, and is only a mode of extended. — Noam Chomsky, Language and problems of knowledge: the Managua lectures, p. 144So matter is whatever physics studies and the object of study of physics is matter: there is no independent general definition of matter, apart from its fitting into the methodology of measurement and controlled experimentation.
In sum, the boundaries between what constitutes matter and everything else remains as vague as the of delimiting science from everything else.In the 19th century, following the development of the, and of, were seen as being the fundamental constituents of matter; atoms formed and.The common definition in terms of occupying space and having mass is in contrast with most physical and chemical definitions of matter, which rely instead upon its structure and upon attributes not necessarily related to volume and mass. At the turn of the nineteenth century, the knowledge of matter began a rapid evolution.Aspects of the Newtonian view still held sway. Discussed matter in his work Matter and Motion. He carefully separates 'matter' from space and time, and defines it in terms of the object referred to in.However, the Newtonian picture was not the whole story. In the 19th century, the term 'matter' was actively discussed by a host of scientists and philosophers, and a brief outline can be found in Levere.
A textbook discussion from 1870 suggests matter is what is made up of atoms:Three divisions of matter are recognized in science: masses, molecules and atoms.A Mass of matter is any portion of matter appreciable by the senses.A Molecule is the smallest particle of matter into which a body can be divided without losing its identity.An Atom is a still smaller particle produced by division of a molecule.Rather than simply having the attributes of mass and occupying space, matter was held to have chemical and electrical properties. In 1909 the famous physicist (1856–1940) wrote about the 'constitution of matter' and was concerned with the possible connection between matter and electrical charge.There is an entire literature concerning the 'structure of matter', ranging from the 'electrical structure' in the early 20th century, to the more recent 'quark structure of matter', introduced today with the remark: Understanding the quark structure of matter has been one of the most important advances in contemporary physics. In this connection, physicists speak of matter fields, and speak of particles as 'quantum excitations of a mode of the matter field'. And here is a quote from de Sabbata and Gasperini: 'With the word 'matter' we denote, in this context, the sources of the interactions, that is (like and ), which are believed to be the fundamental components of matter, or, like the, which are used to introduced mass in a (and that, however, could be composed of more fundamental fermion fields).' In the late 19th century with the of the, and in the early 20th century, with the of the, and the birth of, matter was seen as made up of electrons, and interacting to form atoms. Today, we know that even protons and neutrons are not indivisible, they can be divided into, while electrons are part of a particle family called.
Both are, and are currently seen as being the fundamental constituents of matter.These quarks and leptons interact through four:,. The of particle physics is currently the best explanation for all of physics, but despite decades of efforts, gravity cannot yet be accounted for at the quantum level; it is only described by (see and ). Interactions between quarks and leptons are the result of an exchange of (such as ) between quarks and leptons. The force-carrying particles are not themselves building blocks.
As one consequence, mass and energy (which cannot be created or destroyed) cannot always be related to matter (which can be created out of non-matter particles such as photons, or even out of pure energy, such as kinetic energy). Force carriers are usually not considered matter: the carriers of the electric force (photons) possess energy (see ) and the carriers of the weak force are massive, but neither are considered matter either. However, while these particles are not considered matter, they do contribute to the total mass of atoms, and all systems that contain them. SummaryThe modern conception of matter has been refined many times in history, in light of the improvement in knowledge of just what the basic building blocks are, and in how they interact.The term 'matter' is used throughout physics in a bewildering variety of contexts: for example, one refers to ', 'elementary matter', ' matter, ' matter, '-matter, ' matter, and ' matter. In discussions of matter and, normal matter has been referred to by as koinomatter (Gk.
Common matter). It is fair to say that in, there is no broad consensus as to a general definition of matter, and the term 'matter' usually is used in conjunction with a specifying modifier.The history of the concept of matter is a history of the fundamental length scales used to define matter. Different building blocks apply depending upon whether one defines matter on an atomic or elementary particle level. One may use a definition that matter is atoms, or that matter is, or that matter is leptons and quarks depending upon the scale at which one wishes to define matter.These quarks and leptons interact through four:,. The of particle physics is currently the best explanation for all of physics, but despite decades of efforts, gravity cannot yet be accounted for at the quantum level; it is only described by (see and ).
- Matter(noun)that of which anything is composed; constituent substance; material; the material or substantial part of anything; the constituent elements of conception; that into which a notion may be analyzed; the essence; the pith; the embodiment
- Matter(noun)that of which the sensible universe and all existent bodies are composed; anything which has extension, occupies space, or is perceptible by the senses; body; substance
- Matter(noun)that with regard to, or about which, anything takes place or is done; the thing aimed at, treated of, or treated; subject of action, discussion, consideration, feeling, complaint, legal action, or the like; theme
- Matter(noun)that which one has to treat, or with which one has to do; concern; affair; business
- Matter(noun)affair worthy of account; thing of consequence; importance; significance; moment; -- chiefly in the phrases what matter ? no matter, and the like
- Matter(noun)inducing cause or occasion, especially of anything disagreeable or distressing; difficulty; trouble
- Matter(noun)amount; quantity; portion; space; -- often indefinite
- Matter(noun)substance excreted from living animal bodies; that which is thrown out or discharged in a tumor, boil, or abscess; pus; purulent substance
- Matter(noun)that which is permanent, or is supposed to be given, and in or upon which changes are effected by psychological or physical processes and relations; -- opposed to form
- Matter(noun)written manuscript, or anything to be set in type; copy; also, type set up and ready to be used, or which has been used, in printing
- Matter(verb)to be of importance; to import; to signify
- Matter(verb)to form pus or matter, as an abscess; to maturate
- Matter(verb)to regard as important; to take account of; to care for
Origin: [OE. matere, F. matire, fr. L. materia; perh. akin to L. mater mother. Cf. Mother, Madeira, Material.]


